
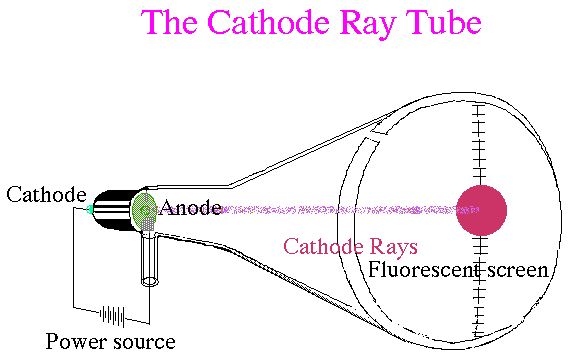
By doing so, he discovered that he could measure the ratio of the mass of a particle to its electric charge. Thomson, the Cavendish Laboratory, and Thompsons cathode ray tube and positive ray apparatus, see J. In the third experiment, Thomson wanted to find if he could determine the basic properties of the particles. He was unsure if the particles were gases, atoms or matter in a finer state of subdivision. Thomson then went on to extract all gases from the cathode ray tube to try and identify all particles in the experiment. All attempts failed when he tried to use the bent rays. He concluded that there was no way to separate the rays and that they needed to be stuck together to function properly.

Thomson found that by bending the rays, very little electric charge was able to pass through the cylinders. Thomson wanted to see if he could separate the electrical charge from the rays, and he also wanted to see if the electric charges were negative or positive.ĭuring his first experiment, he found that when the rays entered the cylinders, they released mostly negative charges. It was used for sending and measuring electric charges. Thomson built a cathode ray tube by putting two cylinders together and sending a voltage through them. He died in Cambridge on August 30, 1940, and is buried in Westminster Abbey near two other influential scientists: Isaac Newton and Charles Darwin.
#Jj thomson cathode ray tube archive#
He left research in 1918 to become Master of Trinity College. Download Atomic & Nuclear Research, 1897-1939 J J Thomsons cathode ray tube with magnet coils, 1897 from Bridgeman Images archive a library of millio. In addition to being awarded the Nobel Prize in 1906, he was knighted in 1908 by King Edward VII. Thomson published 13 books and more than 200 papers in his lifetime. They had one daughter, Joan, and one son, George Paget Thomson, who went on to become a physicist and win a Nobel Prize of his own. Thomson married Rose Paget, one of his students, in 1890. From the cathode ray tube experiments concluded that. He measured the charge and mass of the particles. First, Thomson determined the speed of the rays in the cathode ray tube by passing the beam through crossed electric and magnetic fields. This version of the cathode ray tube, known as. He observed a flow of identical negatively charged particles, irrespective of the gases present in the cathode tube. JJ Thomson giving a lecture demonstration with the Braun cathode ray tube. This was the first use of mass spectrometry. Thomson passed a very high electric charge in a vacuum tube containing different gases. In doing so, he discovered that neon was composed of two different kinds of atoms, and proved the existence of isotopes in a stable element. This led to one of his other famous discoveries in 1912 when he channeled a stream of ionized neon through a magnetic and an electric field and used deflection techniques to measure the charge to mass ratio.
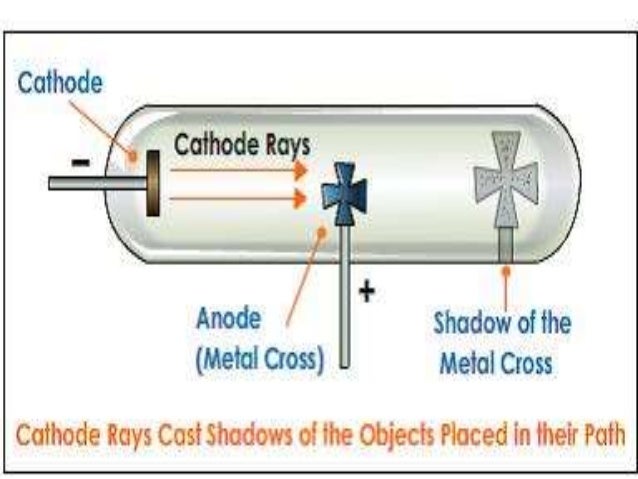
In 1906, Thomson began studying positively charged ions, or positive rays. Important Dates of His Studies May to June 1897-Experimented with a cathode ray tube April 30th, 1897-discovered the electron 1904- suggested the model of the atom was a sphere 1906- Won a Nobel prize How The Electron Was Discovered.
